Innovative treatment helps family severely burned when Hamas set their home on fire
Ariel and Ellay Golan and baby Yael were in life-threatening danger after surviving in their safe room on Oct 7. Pioneering enzyme-based treatments put them on path to recovery
Renee Ghert-Zand is the health reporter and a feature writer for The Times of Israel.

On the morning of October 7, Ariel and Ellay Golan and their 18-month-old daughter Yael hid in the safe room of their home in Kibbutz Kfar Aza as rocket sirens went off incessantly.
As they texted with family members elsewhere in Israel about hearing gunshots outside, they had no idea that terrorists had infiltrated not only their kibbutz but also much of southern Israel. The thousands of terrorists murdered 1,200 people, mainly civilians, and took 240 hostage to Gaza.
The terrorists set fire to their home with them inside during the hours-long rampage. While the Golans survived the onslaught, they were severely injured and taken by helicopter to the National Burns Center at Sheba Medical Center near Tel Aviv.
When The Times of Israel first reported on the young family on October 18, they were fighting for their lives at Sheba. They have now recovered to varying degrees.
“[Father] Ariel and [baby] Yael have been discharged to home recovery while [mother] Ellay is now receiving rehabilitative care at Sheba,” reported Prof. Josef Haik, director of the National Burns Center, in early January.
According to Haik, the positive outcome is due in part to having been treated immediately upon arrival at Sheba with a product called NexoBrid. Developed by Israeli company MediWound and approved by the FDA, it makes treating severe burns easier and quicker. Dozens of October 7 victims have been treated with the product.

Although NexoBrid is approved and sold in 44 countries, MediWound notified foreign clients immediately after October 7 that product supply to Israeli hospitals was the company’s immediate priority and that international shipments would be delayed.
“The IDF and the five burn centers at Israeli hospitals bought 100% of our inventory,” said MediWound CEO Ofer Gonen.
NexoBrid essentially enables trained medical staff to perform enzymatic surgery rather than standard surgery to remove eschar, or dead tissue within a wound, including a severe burn.
The product, used to date on 14,000 patients globally, mainly civilians, comes as a powder in one container and a gel in another. The two are mixed and applied with a tongue depressor and then eventually removed.
“The whole process can be done in one day rather than days and there is less scarring, which is especially important for young people,” Gonen said.
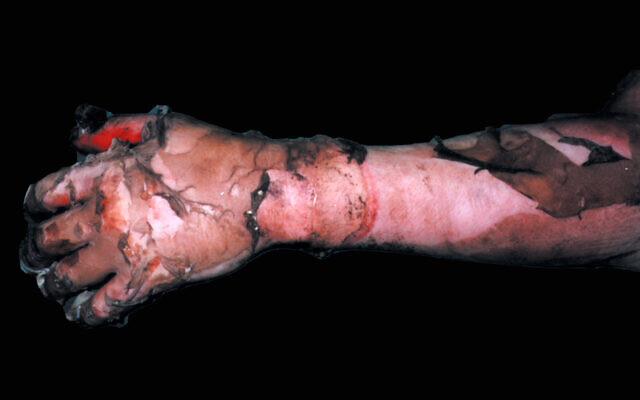
MediWound founder plastic surgeon Lior Rosenberg noted that NexoBrid saves hands and feet, which are the body parts most likely to be exposed and burned as a person defends himself or tries to escape from flames.
“An escharotomy [emergency surgery to remove eschar] must be done to relieve pressure that builds up to save the hands and feet. Our product allows for this to be done without having to cut through burned skin,” he said.
Haik, who has treated burn victims all over the world, said NexoBrid is a game-changer.
“If we hadn’t used it, [the Golans] would have had lots of skin grafts, surgeries, a longer hospitalization, and experienced more pain. By using NexoBrid we had to perform less debridements and saved tissue that otherwise we would have cut off,” he said.
The United States has bought $20 million worth of NexoBrid for its strategic national stockpile. At the end of 2023, the US Department of Defense awarded MediWound an additional $6.7 million toward a total $14.4 million budget for a different version of NexoBrid that can be used on the battlefield.
“The drug as it is now must be stored between two and eight degrees Celsius. The US government wants a stable preparation that can be used in combat situations far from medical facilities or out at the scene of a mass casualty event,” Gonen explained.
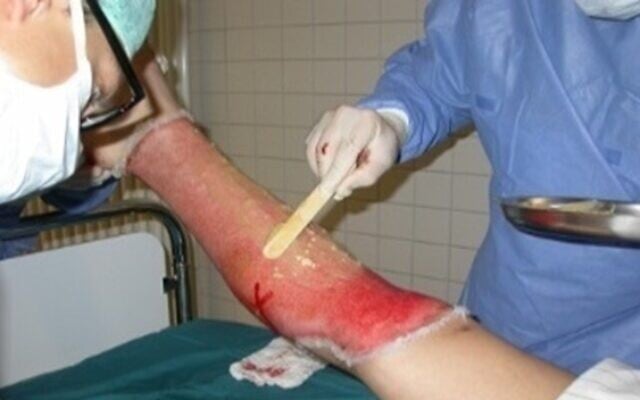
“Fortunately in Israel, we can use the original preparation because we can evacuate burn victims quickly to one of our burn centers,” he said.
According to Gonen, MediWound has received a request from Israel for emergency stockpiling of NexoBrid. This may be possible down the road, but not now.
“MediWound is currently committed to fulfilling obligations in other regions, including the US and Japan. The company’s manufacturing facility [in Yavne] is fully occupied around the clock to ensure the availability of supplies for patients in urgent need,” Gonen said.
The Times of Israel Community.











