Tel Aviv University study augurs hope for pancreatic cancer patients
Researchers find genomic pattern typical of longer-term survivors; develop nanoparticle ‘taxi’ that delivers tumor suppressors straight to cancer tissue
Shoshanna Solomon was The Times of Israel's Startups and Business reporter
A Tel Aviv University study that set out to find what makes some pancreatic cancer patients more likely to survive the deadly disease than others hopes to use the results to develop an effective cocktail of drugs to fight the aggressive cancer as well as other types, researchers said.
The study, which was published Tuesday in Nature Communications, was led by Prof. Ronit Satchi-Fainaro, chair of the Department of Physiology and Pharmacology at TAU’s Sackler Faculty of Medicine.
Pancreatic cancer is among the most aggressive cancers known today. The overwhelming majority of pancreatic cancer patients succumb within a year of diagnosis.
“Despite all the treatments afforded by modern medicine, some 75 percent of all pancreatic cancer patients die within 12 months of diagnosis, including many who die within just a few months,” Satchi-Fainaro said.
“But around 7% of those diagnosed will survive more than five years. We sought to examine what distinguishes the survivors from the rest of the patients,” she said. “We thought that if we could understand how some people live several years with this most aggressive disease, we might be able to develop a new therapeutic strategy.”

The research team examined pancreatic cancer cells and discovered an inverse correlation between a gene that promotes the development of cancer and a cancer suppressor. The levels of miR-34a, a tumor suppressant, were low in pancreatic cancer mouse models and human cell models, while the levels of PLK1, a known oncogene that boosts development of the cancer cells, were high. However, patients who beat the odds — the so-called long-term survivors — had a completely opposite genetic makeup: they had higher levels of the tumor suppressant and lower levels of the PLK1 gene.
The researchers validated their findings with human samples at the Sheba Medical Center, Tel Hashomer in Ramat Gan and then with a bigger cohort of samples at the University of Maryland. An RNA profiling and analysis of samples taken from pancreatic cancer patients showed the same genomic pattern found earlier in mouse and human models of pancreatic cancer.
“This gave us a clear picture of targets for therapy development,” Satchi-Fainaro said in a phone interview. “The idea is to try to ‘re-educate’ the pancreatic cells so as to inhibit the PLK1 gene and bring out, help express, the miR-34a gene,” with the aim of delaying development of the disease and prolong the life of patients.
Suppressing the bad, boosting the good
In a second stage of the research, the scientists devised a new nanoparticle that can selectively deliver genetic material to a tumor, without side effects for the surrounding healthy tissues.
“We designed the nanocarrier to deliver two passengers: the miR-34a, and a small interfering RNA (siRNA) that silences the PLK1,” Satchi-Fainaro said. “The smart nanoparticle knows how to find the pancreatic cancer and release its cargo that will silence the PLK1 and help express the miR-34a.”
“The nanoparticle is like a taxi carrying two important passengers,” she explained. “Many oncology protocols are cocktails, but the drugs usually do not reach the tumor at the same time. But our ‘taxi’ kept the ‘passengers’ — and the rest of the body — safe the whole way, targeting only the tumor tissue. Once it ‘parked,’ an enzyme present in pancreatic cancer caused the carrier to biodegrade, allowing the therapeutic cargo to be released at the correct address — the tumor cells.”
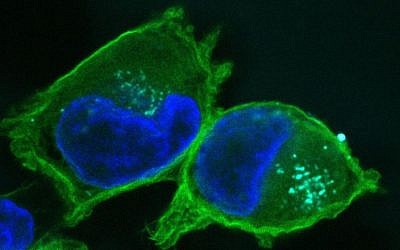
To validate their findings, the scientists injected the new nanoparticles into pancreatic tumor-bearing mice and observed that by re-balancing these two targets — increasing the expression of one and blocking that of the other — they significantly prolonged the survival of the mice.
“We managed to change the genetic signature of pancreatic cancer cells and inhibit the cancer gene and increase the survival of mice,” she said. “Hopefully, this will lead to the development of new drugs that will increase the overall survival of cancer patients.”
The study was also conducted by Hadas Gibori and Dr. Shay Eliyahu, both of Satchi-Fainaro’s multidisciplinary laboratory, in collaboration with Prof. Eytan Ruppin of TAU’s Computer Science Department and the University of Maryland and Prof. Iris Barshack and Dr. Talia Golan of Sheba Medical Center, Tel Hashomer.
Research for the study was funded by the European Research Council, Tel Aviv University’s Cancer Biology Research Center and the Israel Science Foundation.
The Times of Israel Community.












