Fusion of quantum dots leads to new world of artificial materials
Hebrew University nanoscientists say their manipulation of these artificial atoms could be used for new kinds of enhanced TV monitors or new color-based markers in biology
Shoshanna Solomon was The Times of Israel's Startups and Business reporter
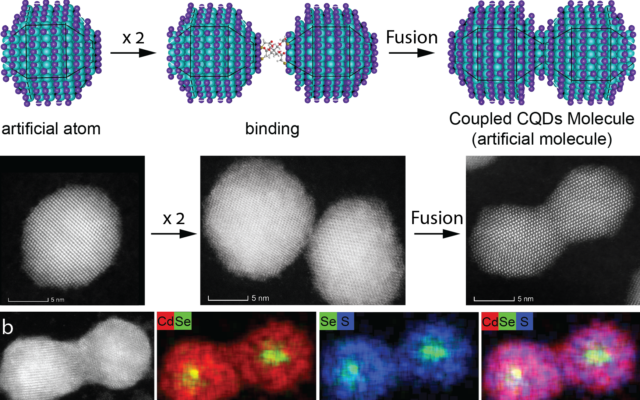
A team of Israeli scientists say they are able to create a whole new world of artificial materials by playing with the composition of quantum dots, tiny artificial atoms that are created chemically in a lab.
The nanoscience researchers, led by Professor Uri Banin at the Hebrew University of Jerusalem’s Institute of Chemistry and Center for Nanoscience and Nanotechnology, took the quantum dots and fused them together, creating new artificial molecules, which can be used as the basis for a whole new variety of materials.
Their findings were published in the latest edition of Nature Communications.
These artificial molecules can be compared to the rich selection of molecules that can be created from the elements of the periodic table. The periodic table of elements was set out in 1869 by Russia’s Dmitri Mendeleev, who classified 118 atoms found in nature according to their chemical properties. These atoms can be combined to form 150 million original molecules.

The Israeli researchers said they were “reinventing” the concept of the periodic table, this time with artificial atoms.
“This is an important discovery in the field of nano-particles and for the discovery of new nano-materials” that could have a variety of applications, said Banin in a phone interview.
In the past twenty years, scientists’ understanding of the physical properties of quantum dots and their levels of control over these tiny particles have increased tremendously, the Hebrew University said in a statement on Monday.
This has led to widespread application of quantum dots — from bio-imaging and bio-tracking, relying on the fact that quantum dots emit different colors based on their size, to solar energy and next-generation TV monitors with exceptional color quality.
The Hebrew University team’s discovery could lead to new kinds of enhanced displays, Banin said, or could be used as new color-based markers in biology.
“With these artificial atoms, we can enrich the variety of materials,” he said.
Quantum dots are nano-sized chunks of crystal, created through a chemical process in labs, that contain hundreds to thousands of semiconductor atoms. When viewed through an electron microscope they look like dots.
As with real atoms, when you combine artificial atoms, they create a new (artificial) molecule with unique properties and characteristics. These molecules are referred to as “artificial” because they’re not one of the 150 million original molecules that have been formed by combining atoms from the 118 known elements in the periodic table.
Unlike their periodic table counterparts, quantum dot atoms are “mercurial” in nature, changing their physical, electronic and optical properties when their size changes.
For example, a larger quantum dot will emit a red light, while a smaller one of the same material will emit a green light.
This new development lays the foundations for the formation of a wide variety of fused quantum dot molecules.
Taking into account the “rich selection of size and composition” of these quantum dots, there are many “exciting possibilities” for the creation of new artificial molecules, with a “great promise for their utilization in numerous opto-electronic, sensing and quantum technologies applications,” Banin said in the statement.
The Times of Israel Community.











