Stem cell treatment ‘freezes’ MS, lets some walk again, Jerusalem study finds
Small study saw improvement in 73% of multiple sclerosis patients who got spinal injection containing treatment made from their own cells; larger trials now planned
Nathan Jeffay is The Times of Israel's health and science correspondent
Clinical trials for a new stem cell treatment have “frozen” the progression of multiple sclerosis, and left some patients in better condition than before they were injected, even ditching walking sticks, according to Jerusalem doctors.
The biopharmaceutical company NeuroGenesis has licensed a method developed at Hadassah Medical Center to take stem cells from MS patients’ own bone marrow.
“We inject it, and it rebuilds the external lining of the nerves, the myelin, that is affected by multiple sclerosis,” Dimitrios Karussis, head of Hadassah’s MS department and one of the neurologists behind the innovation, told The Times of Israel.
The outcome of the Phase 2 clinical trial was conducted at Hadassah and published last month in Brain, a peer-reviewed journal published by Oxford University. NeuroGenesis CEO Tal Gilat said it “demonstrated unprecedented results in disease improvement and repair, which is an unmet need for over 500,000 progressive MS patients in the US alone.”
“This opens a window for a completely new approach to multiple sclerosis,” said Karussis, who headed the trial, along with Dr. Petrou Panayiota and Dr. Ibrahim Kassis. “We hope that, alongside the use of other medications, this can lead to a breakthrough with this disease.”
He added: “Although we currently have several good treatment options for MS in patients who experience remissions, in patients who have continuous progression toward disability, we fall short in providing effective treatment that could substantially suppress the progression of disability.”
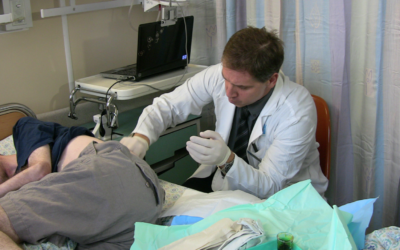
The trial involved 48 patients with progressive MS, who had unsuccessfully tried two drugs. A third were given a placebo, a third got a regular intravenous injection, and a third had the stem cells injected directly into the central nervous system, through the cerebrospinal fluid.
By the end of a one-year, three-injection course of treatment, MS progression stopped in two-fifths of the patients who received the stem cells intravenously. Among those who received the cells in the central nervous system, disease progression was frozen in 60% of cases, according to Karussis.

During the second half of the trial, some 73% of the patients who were receiving injections to the central nervous system showed signs of improvement in a combined disability measurement that assesses ability to walk, move arms, cognitive function and brain function as documented by neurological examination.
“Some patients stopped using a walker or a stick, and some others increased the distance they can walk,” said Karussis.
He is hopeful the next phase of trials could be completed within three years, paving the way for the stem cells to be offered through hospitals worldwide, but acknowledged that more research is needed. He commented: “We are certainly enthusiastic but when the trial is relatively small we need to confirm this data with large Phase 3 trials.”
Gilat said that the company has been holding discussions with America’s Food and Drug Administration and expects to initiate a Phase 3 trial spanning the US, Israel, and Europe in 2021.
The Times of Israel Community.











