In first, Haifa man regains his vision after getting artificial cornea implant
Jamal Furani, 78, is the first person implanted with CorNeat’s product; after operation, he could read text and recognize family members, startup says
Shoshanna Solomon was The Times of Israel's Startups and Business reporter
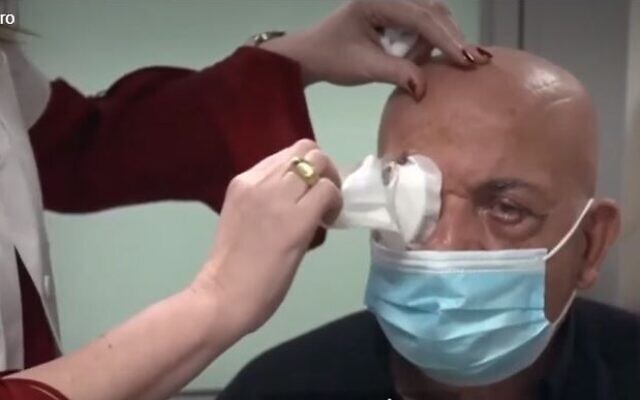
A 78-year-old blind man, Jamal Furani from Haifa, regained his sight after he was implanted with an artificial cornea developed by CorNeat Vision, the biomimetic implant firm said. It was the first such successful implant.
The artificial cornea, which fits into the eye wall, was implanted earlier this month by Prof. Irit Bahar, director of the Ophthalmology Department at Rabin Medical Center in Israel. Upon removal of bandages, Furani was able to read text and recognize family members, the startup said in a statement earlier this month.
Furnai, who suffered from edema and other diseases, had a damaged cornea, causing a decade of vision loss and defining him as legally blind.
CorNeat’s product, made out of 100% synthetic, non-degradable porous material, replaces scarred or deformed corneas and requires no donor tissue. It uses cell technology and nanoscale chemical engineering to mimic the existing cell environment. When implanted, the material integrates with the live tissue and stimulates cellular proliferation, leading to even more integration. The implant thus does not trigger an adverse immune system response, the company said.

Corneal diseases account for the second leading cause of blindness in most developing world countries, and the World Health Organization estimates that some 2 million patients a year suffer from corneal blindness.
“Unveiling this first implanted eye and being in that room, in that moment, was surreal,” said Dr. Gilad Litvin, CorNeat Vision’s co-founder, chief medical officer, and the inventor of the CorNeat KPro. “After years of hard work, seeing a colleague implant the CorNeat KPro with ease and witnessing a fellow human being regain his sight the following day was electrifying and emotionally moving, there were a lot of tears in the room. This is an extremely important milestone for CorNeat Vision.”
The implantation process is fairly simple and takes less than an hour, the company said in a YouTube video.
A total of 10 patients are approved for the clinical trial at Rabin Medical Center in Israel with two additional sites planned to open this month in Canada and six others at different stages in the approval process in France, the US, and the Netherlands, said Almog Aley-Raz, CorNeat Vision’s co-founder, chief executive officer and VP R&D, in the statement.
The first trial includes blind patients who are not suitable candidates for- or have failed one or more corneal transplantations, he added.
“Given the exceptional visual performance of our device, the expected healing time and retention,” he said, the firm plans to start a second study later this year with broader indications to approve the firm’s artificial cornea “as a first line treatment, displacing the use of donor tissue used in full thickness corneal transplantations.”
Founded in 2015 and based in Ra’anana, CorNeat has also developed the CorNeat EverPatch, a synthetic non-degradable scleral patch, and the CorNeat eShunt, a glaucoma drainage device. According to the database of Start-Up Nation Central, the firm has raised $12.3 million from investors to date.
The Times of Israel Community.











