Israeli startup develops device to jumpstart digestive system
E-Motion Medical’s tube delivers electrical stimulation to the esophagus; having won EU regulatory nod, it hopes for Israeli and US approval soon
Shoshanna Solomon was The Times of Israel's Startups and Business reporter

Israeli medical devices startup E-Motion Medical Ltd. has developed a way to restore digestive movement in patients who, due to trauma, neurological dysfunction or old age, have lost the ability to swallow.
The company’s patented technology — a tube that can be inserted into patients like a feeding tube — delivers unique patterns of electrical stimulation to the esophagus, creating contractions of the muscle in a variety of locations along the esophageal body that mimic the natural lower esophageal swallowing motions in a healthy person.
The system restores the natural motor function of the digestive system for patients with acute gastrointestinal dysmotility (GID).
Acute GID is common in neurological, trauma, surgical, geriatric and severely diabetic patients. It manifests itself as reduction in, or lack of, motility in the digestive system, and leads to increased risk of complications and longer recovery time.
“There is currently no adequate solution available for the widespread problem of acute digestive dysmotility. Our technology provides a new therapy to promote the physical function of the digestive system, speeds up recovery and consequently lowers the financial burden on the healthcare system,” said Amichay Gross, co-founder and CEO of E-Motion Medical, in a phone interview.
After a trauma or surgery, or if a patient is in coma or in an induced coma, the digestive system shuts down. The electric stimulation generated by E-Motion’s tube has several positive effects, including pushing down gastric content and preventing it from refluxing up the esophagus, increased gastric emptying, and helping push food down to the intestine for it to be digested and absorbed as nutrients that the critically ill body needs for healing.
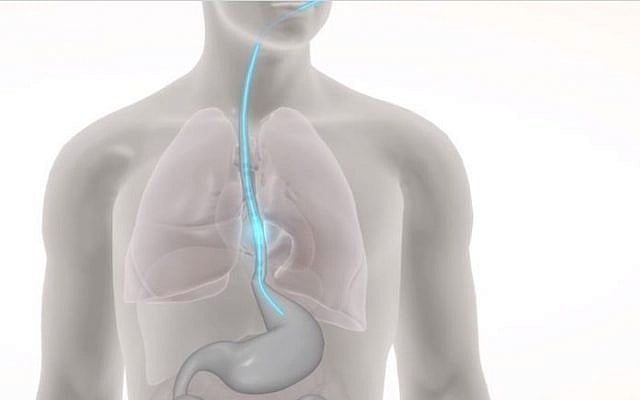
“Our system wakes up the gut, and does something like physical therapy for the esophagus,” Gross said. “Just as a ventilator keeps the lungs functioning and helps restore their natural functioning, our system keeps the esophagus in action and helps it get back on track sooner.”
Last month, E-Motion Medical got the regulatory nod from European authorities to start marketing the device in the EU.

“The CE Mark is an important milestone for E-Motion Medical, as it will allow commercial access to hospitals across Europe,” he said.
The company hopes to get permits from the Israeli regulator within two months and from the US authorities by mid-2019, he said.
To date, the E-Motion System has been tested on approximately 230 people in four clinical trials conducted in 12 hospitals in Europe and Canada.
The latest trial was a randomized, double-blind trial of 155 patients. This study demonstrated that the treatment provided by the E-Motion System promotes gastric motility, reduces risk of aspirations, and speeds up recovery.
The firm was set up in 2011 by Michael Tal and Gross.
The Times of Israel Community.











