Israeli tech to help pathologists detect cancer gets FDA ‘breakthrough’ nod
Designation is ‘important step’ toward startup Ibex Medical Analytics making its software available in US
Shoshanna Solomon was The Times of Israel's Startups and Business reporter
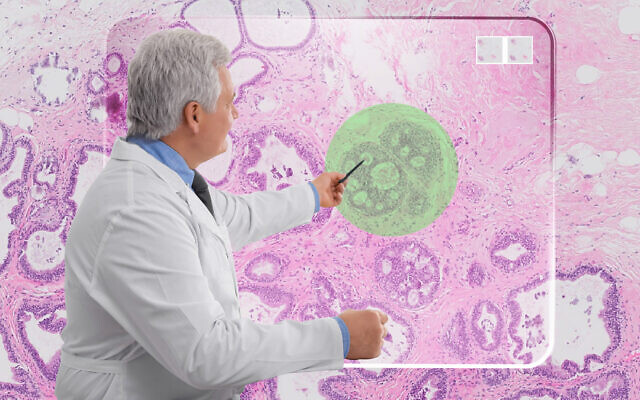
Ibex Medical Analytics, the maker of AI-based cancer diagnostic software, said Wednesday it has received a breakthrough device designation by the US Food and Drug Administration, which will help the startup fast-track the clinical review and regulatory approval of its technology. The FDA nod is an important step in making the technology broadly available in the US, the company said.
The FDA’s breakthrough device designation is granted to technologies that have the potential to provide more effective treatment or diagnosis of life-threatening diseases, such as cancer. The designation enables the company to work in close collaboration with the FDA and get a review of its technology. It also provides formal acknowledgement of the platform’s potential benefit to help pathologists in their work to detect and diagnose cancer, the company said.
Ibex’s Galen software uses AI-based algorithms to help pathologists analyze biopsies and improve the quality of cancer diagnosis, implement real-time quality control, reduce diagnosis time and boost productivity. The software is already deployed in labs worldwide where it is used as part of everyday clinical practice, and its positive outcomes have been demonstrated in clinical studies, Ibex said in a statement.
“Oncology treatments have made great strides, but in order to save more lives it is also essential to see technological advances in cancer diagnostics,” said David Shulkin, former secretary, US Department of Veterans Affairs and adviser to Ibex. “Enhancing the accuracy of cancer diagnosis and improving the efficiency for the pathologist is paramount to improving quality and affordability of cancer care. Ibex’s AI platform has demonstrated success in helping pathologists worldwide improve care for patients with cancer. This FDA designation is an important step forward in making this technology broadly available in the United States.”
The Times of Israel Community.