 From the Liveblog of
Wednesday, February 3, 2021
From the Liveblog of
Wednesday, February 3, 2021
UK says study vindicates choice to delay 2nd vaccine dose
Britain’s health chief says that a new study suggesting that a single dose of the Oxford-AstraZeneca COVID-19 vaccine provides a high level of protection for 12 weeks supports the government’s strategy of delaying the second shot so it can protect more people quickly with a first dose.
Britain’s decision has been criticized as risky by other European countries, but Health Secretary Matt Hancock says the study “backs the strategy that we’ve taken and it shows the world that the Oxford vaccine works effectively.”
Hancock’s comments comes after Oxford University released a study showing the vaccine cut transmission of the virus by two-thirds and prevented severe disease.
Mene Pangalos, executive vice president of biopharmaceuticals research and development at AstraZeneca, says no patients experienced severe disease or hospitalization three weeks after receiving a first dose, and that efficacy appeared to increase up to 12 weeks after the initial shot.
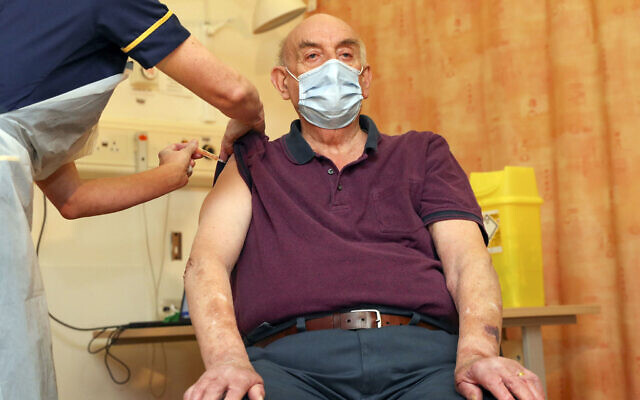
“Our data suggest you want to be as close to the 12 weeks as you can” for the second dose, he tells a news conference.
The study has not been peer-reviewed yet and does not address the efficacy of the Pfizer-BioNTech vaccine, the other one currently in use in the UK. Pfizer recommends that its shots be given 21 days apart and has not endorsed the UK government’s decision to lengthen the time between doses.
But the Oxford research was greeted with excitement by UK officials under pressure to justify their decision to delay the second dose.
The Times of Israel Community.











