Pfizer says vaccine 100% effective for 12-15 olds, asks for US authorization
Vaccine maker says it plans to follow up with requests to regulators in other countries in coming days; says shot ‘well tolerated’ by young people
WASHINGTON — Pfizer-BioNTech asked for authorization Friday to use their COVID-19 vaccine on 12-15 year olds in the United States.
The companies said in a statement that they plan to make similar requests of other regulatory authorities worldwide in coming days.
Their request to the Food and Drug Administration in the US comes after Phase 3 clinical trials of the Pfizer vaccine in 12-15 year olds were 100 percent effective in warding off the disease, according to the companies.
In late March they published the results of trials carried out with 2,260 adolescents in the US who the companies said showed “robust anti-body responses.”
The vaccine was “well tolerated with side effects generally consistent with those observed in participants 16 to 25 years of age,” the companies said Friday.
For now the vaccine has emergency authorization for use in people 16 and older.
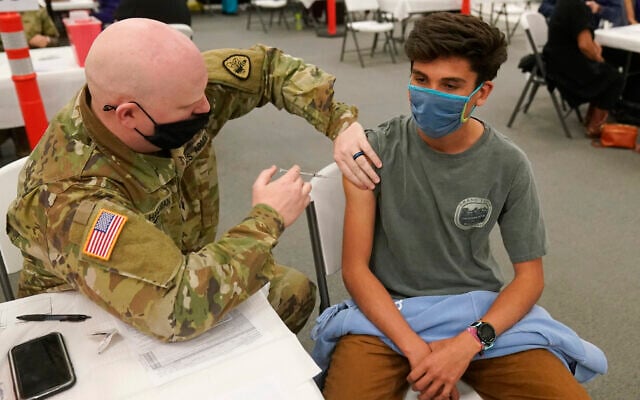
Children are less likely to develop severe COVID so vaccinating them has been less of a priority than getting older people inoculated.
The BioNTech/Pfizer shot is based on novel mRNA technology and was the first COVID-19 vaccine to be approved in the West late last year.
Israel has so far vaccinated some 600 children between the ages of 12 and 16 in at-risk groups against the coronavirus with the Pfizer-BioNTech vaccine and has seen no significant side effects from the shot, a top health official said last month.
“We have so far immunized somewhere around 600 children,” Dr. Boaz Lev, a top member of the Health Ministry’s advisory team to the cabinet, told the Guardian. “We didn’t see any major side effects; even minor ones are quite rare. This is encouraging.”
Prime Minister Benjamin Netanyahu has said children will soon begin receiving vaccines, but didn’t specify a timeline.
The Israeli children who have been approved by medical authorities for vaccination had known risk factors including obesity, diabetes, severe lung and heart disease, immunosuppression disorders and cancer.

The Health Ministry has recommended vaccinating some teenagers age 12-15 against COVID-19 if they suffer from specific underlying conditions. The report did not say whether all the children vaccinated in Israel were over the age of 12.
The Times of Israel Community.











